Empirical Formula For Ascorbic Acid
3.v: Empirical Formulas from Analysis
- Page ID
- 21710
- To empathise the definition and difference between empirical formulas and chemical formulas
- To understand how combustion analysis can exist used to identify chemical formulas
Chemical formulas tell you how many atoms of each element are in a compound, and empirical formulas tell you the simplest or nearly reduced ratio of elements in a chemical compound. If a compound'due south chemic formula cannot exist reduced any more, so the empirical formula is the same as the chemical formula. Combustion analysis tin determine the empirical formula of a chemical compound, just cannot determine the chemical formula (other techniques tin can though). Once known, the chemical formula can be calculated from the empirical formula.
Empirical Formulas
An empirical formula tells us the relative ratios of unlike atoms in a compound. The ratios hold true on the tooth level likewise. Thus, H2O is composed of ii atoms of hydrogen and 1 atom of oxygen. Besides, ane.0 mole of HtwoO is composed of 2.0 moles of hydrogen and i.0 mole of oxygen. We tin can as well work backwards from molar ratios since if we know the molar amounts of each element in a compound nosotros tin determine the empirical formula.
Mercury forms a compound with chlorine that is 73.9% mercury and 26.1% chlorine past mass. What is the empirical formula?
Solution
Let'south say we had a 100 gram sample of this compound. The sample would therefore contain 73.9 grams of mercury and 26.ane grams of chlorine. How many moles of each cantlet do the individual masses represent?
For Mercury:
\[(73.ix \;g) \times \left(\dfrac{1\; mol}{200.59\; g}\correct) = 0.368 \;moles \nonumber \]
For Chlorine:
\[(26.1\; g) \times \left(\dfrac{one\; mol}{35.45\; one thousand}\right) = 0.736\; mol \nonumber \]
What is the tooth ratio between the two elements?
\[\dfrac{0.736 \;mol \;Cl}{0.368\; mol\; Hg} = 2.0 \nonumber \]
Thus, we accept twice as many moles (i.e. atoms) of \(\ce{Cl}\) equally \(\ce{Hg}\). The empirical formula would thus be (remember to list cation first, anion last):
\[\ce{HgCl2} \nonumber \]
Chemic Formula from Empirical Formula
The chemical formula for a chemical compound obtained past limerick assay is always the empirical formula. We tin obtain the chemical formula from the empirical formula if we know the molecular weight of the chemical compound. The chemical formula will always be some integer multiple of the empirical formula (i.e. integer multiples of the subscripts of the empirical formula). The general menstruation for this approach is shown in Effigy \(\PageIndex{ane}\) and demonstrated in Example \(\PageIndex{2}\).

Vitamin C (ascorbic acid) contains 40.92 % C, 4.58 % H, and 54.50 % O, past mass. The experimentally determined molecular mass is 176 amu. What is the empirical and chemical formula for ascorbic acid?
Solution
Consider an arbitrary amount of 100 grams of ascorbic acrid, so we would have:
- 40.92 grams C
- 4.58 grams H
- 54.50 grams O
This would give united states how many moles of each chemical element?
- Carbon
\[ (40.92\; \cancel{thousand\; C}) \times \left( \dfrac{1\; mol\; C}{12.011\; \abolish{m\; C}} \right) = iii.407\; mol \; C \nonumber \]
- Hydrogen
\[ (4.58\; \cancel{g\; H}) \times \left( \dfrac{1\; mol\; H}{1.008\; \abolish{g\; H}} \correct) = 4.544\; mol \;H \nonumber \]
- Oxygen
\[ (54.50\; \cancel{g\; O}) \times \left( \dfrac{1\; mol\; O}{15.999\; \abolish{grand\; O}} \right) = three.406\; mol \; O \nonumber \]
Determine the simplest whole number ratio by dividing by the smallest molar amount (iii.406 moles in this instance - see oxygen):
- Carbon
\[ C= \dfrac{3.407\; mol}{iii.406\; mol} \approx 1.0 \nonumber \]
- Hydrogen
\[ C= \dfrac{four.5.44\; mol}{3.406\; mol} = i.0 \nonumber \]
- Oxygen
\[ C= \dfrac{iii.406\; mol}{3.406\; mol} = 1.0 \nonumber \]
The relative tooth amounts of carbon and oxygen announced to be equal, but the relative molar corporeality of hydrogen is higher. Since we cannot take "fractional" atoms in a compound, we demand to normalize the relative amount of hydrogen to be equal to an integer. i.333 would appear to exist one and 1/iii, so if we multiply the relative amounts of each cantlet by '3', we should be able to become integer values for each atom.
C = (1.0)*iii = 3
H = (1.333)*3 = iv
O = (1.0)*3 = 3
or
\[\ce{C3H4O3} \nonumber \]
This is our empirical formula for ascorbic acid.
What about the chemic formula? We are told that the experimentally determined molecular mass is 176 amu . What is the molecular mass of our empirical formula?
(iii*12.011) + (4*1.008) + (3*fifteen.999) = 88.062 amu
The molecular mass from our empirical formula is significantly lower than the experimentally adamant value. What is the ratio between the two values?
(176 amu/88.062 amu) = 2.0
Thus, information technology would appear that our empirical formula is essentially one half the mass of the actual molecular mass. If we multiplied our empirical formula past '2', then the molecular mass would be correct. Thus, the bodily chemical formula is:
2* C3H4O3 = Chalf-dozenH8Ovi
Empirical Formulas: Empirical Formulas, YouTube(opens in new window) [youtu.exist]
Combustion Analysis
When a chemical compound containing carbon and hydrogen is subject to combustion with oxygen in a special combustion apparatus all the carbon is converted to CO2 and the hydrogen to HiiO (Effigy \(\PageIndex{2}\)). The amount of carbon produced can be adamant by measuring the amount of COtwo produced. This is trapped past the sodium hydroxide, and thus we can monitor the mass of COtwo produced by determining the increase in mass of the CO2 trap. Likewise, we can determine the corporeality of H produced by the amount of H2O trapped by the magnesium perchlorate.
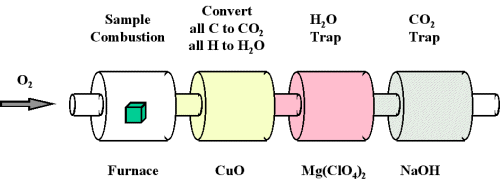
One of the virtually common ways to make up one's mind the elemental composition of an unknown hydrocarbon is an belittling procedure called combustion analysis. A small, carefully weighed sample of an unknown chemical compound that may contain carbon, hydrogen, nitrogen, and/or sulfur is burned in an oxygen atmosphere,Other elements, such as metals, can be determined by other methods. and the quantities of the resulting gaseous products (CO2, H2O, Due north2, and And soii, respectively) are adamant by one of several possible methods. 1 procedure used in combustion analysis is outlined schematically in Effigy \(\PageIndex{3}\) and a typical combustion analysis is illustrated in Examples \(\PageIndex{three}\) and \(\PageIndex{iv}\).

What is the empirical codify for isopropyl alcohol (which contains but C, H and O) if the combustion of a 0.255 grams isopropyl alcohol sample produces 0.561 grams of CO2 and 0.306 grams of HiiO?
Solution
From this information quantitate the corporeality of C and H in the sample.
\[ (0.561\; \cancel{g\; CO_2}) \left( \dfrac{ane \;mol\; CO_2}{44.0\; \cancel{1000\;CO_2}}\right)=0.0128\; mol \; CO_2 \nonumber \]
Since one mole of CO2 is made up of i mole of C and two moles of O, if we have 0.0128 moles of CO2 in our sample, then we know we have 0.0128 moles of C in the sample. How many grams of C is this?
\[ (0.0128 \; \abolish{mol\; C}) \left( \dfrac{12.011\; m \; C}{1\; \cancel{mol\;C}}\correct)=0.154\; g \; C \nonumber \]
How most the hydrogen?
\[ (0.306 \; \abolish{k\; H_2O}) \left( \dfrac{one\; mol \; H_2O}{xviii.0\; \abolish{g \;H_2O}}\right)=0.017\; mol \; H_2O \nonumber \]
Since i mole of H2O is made upward of ane mole of oxygen and two moles of hydrogen, if we accept 0.017 moles of HtwoO, so we have 2*(0.017) = 0.034 moles of hydrogen. Since hydrogen is almost 1 gram/mole, we must take 0.034 grams of hydrogen in our original sample.
When nosotros add together our carbon and hydrogen together we get:
0.154 grams (C) + 0.034 grams (H) = 0.188 grams
But we know nosotros combusted 0.255 grams of isopropyl alcohol. The 'missing' mass must exist from the oxygen atoms in the isopropyl booze:
0.255 grams - 0.188 grams = 0.067 grams oxygen
This much oxygen is how many moles?
\[ (0.067 \; \cancel{thousand\; O}) \left( \dfrac{1\; mol \; O}{15.994\; \cancel{g \;O}}\right)=0.0042\; mol \; O \nonumber \]
Overall therefore, nosotros have:
- 0.0128 moles Carbon
- 0.0340 moles Hydrogen
- 0.0042 moles Oxygen
Separate by the smallest tooth amount to normalize:
- C = iii.05 atoms
- H = eight.1 atoms
- O = 1 atom
Within experimental error, the most likely empirical formula for propanol would exist \(C_3H_8O\)
Naphthalene, the active ingredient in ane diversity of mothballs, is an organic compound that contains carbon and hydrogen simply. Consummate combustion of a 20.10 mg sample of naphthalene in oxygen yielded 69.00 mg of CO2 and eleven.thirty mg of H2O. Determine the empirical formula of naphthalene.
Given: mass of sample and mass of combustion products
Asked for: empirical formula
Strategy:
- Use the masses and molar masses of the combustion products, COii and HtwoO, to calculate the masses of carbon and hydrogen present in the original sample of naphthalene.
- Use those masses and the molar masses of the elements to summate the empirical formula of naphthalene.
Solution:
A Upon combustion, ane mol of \(\ce{CO2}\) is produced for each mole of carbon atoms in the original sample. Similarly, i mol of HiiO is produced for every two mol of hydrogen atoms present in the sample. The masses of carbon and hydrogen in the original sample can be calculated from these ratios, the masses of COtwo and H2O, and their molar masses. Considering the units of molar mass are grams per mole, we must first catechumen the masses from milligrams to grams:
\[ mass \, of \, C = 69.00 \, mg \, CO_2 \times {1 \, g \over chiliad \, mg } \times {1 \, mol \, CO_2 \over 44.010 \, chiliad \, CO_2} \times {1 \, mol C \over 1 \, mol \, CO_2 } \times {12.011 \,thou \over one \, mol \, C} \nonumber \]
\[ = one.883 \times x^{-two} \, thousand \, C \nonumber \]
\[ mass \, of \, H = 11.30 \, mg \, H_2O \times {1 \, g \over 1000 \, mg } \times {1 \, mol \, H_2O \over 18.015 \, g \, H_2O} \times {ii \, mol H \over 1 \, mol \, H_2O } \times {1.0079 \,g \over 1 \, mol \, H} \nonumber \]
\[ = one.264 \times ten^{-3} \, 1000 \, H \nonumber \]
B To obtain the relative numbers of atoms of both elements present, we need to calculate the number of moles of each and separate past the number of moles of the element present in the smallest corporeality:
\[ moles \, C = 1.883 \times ten^{-2} \,g \, C \times {1 \, mol \, C \over 12.011 \, g \, C} = 1.568 \times 10^{-3} \, mol C \nonumber \]
\[ moles \, H = ane.264 \times 10^{-3} \,g \, H \times {1 \, mol \, H \over 1.0079 \, g \, H} = 1.254 \times 10^{-3} \, mol H \nonumber \]
Dividing each number by the number of moles of the element present in the smaller amount gives
\[H: {1.254\times x^{−3} \over 1.254 \times 10^{−three}} = 1.000 \, \, \, C: {1.568 \times 10^{−iii} \over 1.254 \times 10^{−3}}= 1.250 \nonumber \]
Thus naphthalene contains a ane.25:1 ratio of moles of carbon to moles of hydrogen: C1.25H1.0. Because the ratios of the elements in the empirical formula must be expressed as minor whole numbers, multiply both subscripts past 4, which gives C5Hfour every bit the empirical formula of naphthalene. In fact, the chemical formula of naphthalene is CxHviii, which is consistent with our results.
- Xylene, an organic compound that is a major component of many gasoline blends, contains carbon and hydrogen but. Complete combustion of a 17.12 mg sample of xylene in oxygen yielded 56.77 mg of COtwo and fourteen.53 mg of H2O. Determine the empirical formula of xylene.
- The empirical formula of benzene is CH (its chemical formula is Chalf-dozenHhalf dozen). If 10.00 mg of benzene is subjected to combustion analysis, what mass of COtwo and H2O will be produced?
- Answer a
-
The empirical formula is C4H5. (The chemic formula of xylene is actually C8H10.)
- Answer b
-
33.81 mg of CO2; 6.92 mg of H2O
Combustion Assay: Combustion Analysis, YouTube(opens in new window) [youtu.be]
Empirical Formula For Ascorbic Acid,
Source: https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_Chemistry_-_The_Central_Science_(Brown_et_al.)/03%3A_Stoichiometry-_Chemical_Formulas_and_Equations/3.05%3A_Empirical_Formulas_from_Analysis
Posted by: lemendessaimis35.blogspot.com


0 Response to "Empirical Formula For Ascorbic Acid"
Post a Comment